Pipeline
Innovation Platform
 Specialized Brain-Penetrant Drug Development Platform
Specialized Brain-Penetrant Drug Development PlatformAs global populations age, the burden of brain diseases continues to rise, intensifying the need for effective therapies. Yet the brain’s complexity, and the blood–brain barrier (BBB), which blocks more than 98% of drug molecules, makes therapeutic development uniquely challenging. Many promising candidates fail simply because they cannot reach their intended site of action.
Hyperway Pharma has built a specialized, high‑efficiency platform dedicated to overcoming this barrier. Supported by an expert R&D team, robust technical capabilities, and advanced precision instrumentation, the platform integrates molecular design, compound synthesis and screening, pharmacokinetic evaluation, and toxicology research. Its mission is to consolidate resources, strengthen collaborative innovation, and accelerate the development of novel therapeutics capable of effectively penetrating the brain.
This platform supports multiple innovative pipelines spanning primary and secondary brain tumors, autoimmune diseases, and pain. Several neurological candidates have demonstrated brain penetration rates exceeding 60%, and HBW‑3210 capsules have successfully advanced into Phase I clinical trials.

 Differentiated R & D pipeline
Differentiated R & D pipelineHyperway Pharma is addressing one of China’s core innovation challenges: widespread pipeline homogeneity. The company is building a distinctly differentiated R&D engine centered on high‑value therapeutic areas such as pain and oncology, supported by a specialized platform for developing brain‑penetrant therapeutics capable of crossing the blood–brain barrier.
In parallel, Hyperway advances validated, high‑value targets with the ambition to deliver Best‑in‑Class (BIC) or First‑in‑Class (FIC) medicines. By securing early positions in emerging markets and leveraging first‑mover advantages, the company is strategically positioned to capture future opportunities ahead of competitors.
This focused, differentiated pipeline strengthens Hyperway Pharma’s identity as a forward‑looking leader shaping the next generation of innovative drug development.

 Streamlined Drug Discovery and Development
Streamlined Drug Discovery and DevelopmentInnovative drug discovery demands scientific rigor, cross‑disciplinary insight, and the ability to turn breakthroughs into real‑world impact. Hyperway Pharma has built an R&D engine designed for exactly this purpose.
Guided by a globally experienced scientific leadership team and strengthened by a dynamic cohort of post‑80s R&D innovators, we take a disciplined and scientifically rigorous approach to pipeline strategy. Our capabilities span the full innovation continuum, from insight‑driven target selection and innovative drug design to advanced complex‑compound synthesis and a globally competitive CMC platform.
With a high‑velocity clinical development engine at the core, Hyperway Pharma operates an R&D system engineered for both scientific excellence and operational speed, ensuring that promising therapies advance with efficiency and uncompromising quality.

Pipeline Flagship Under Development
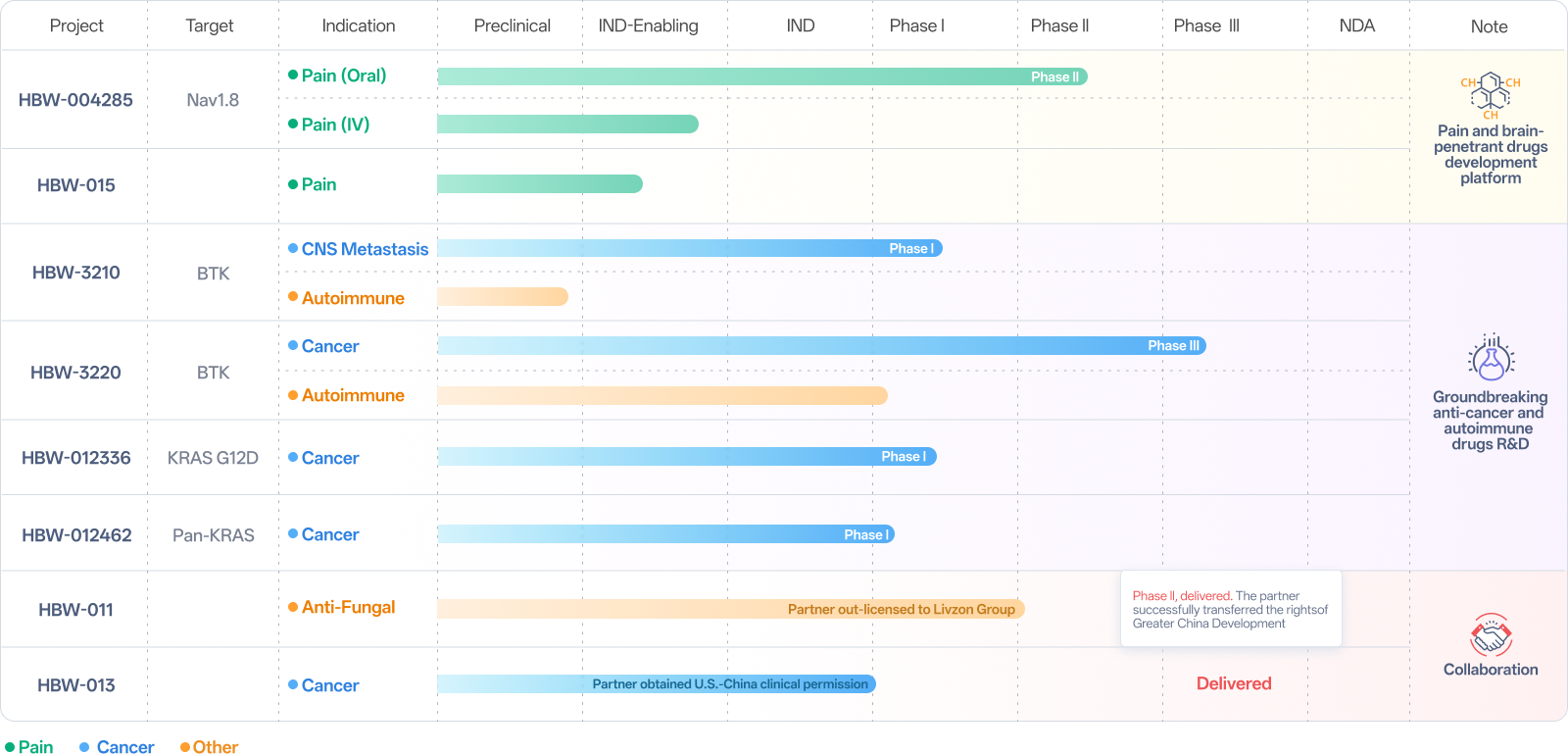
The analgesic drug market presents a vast unmet demand. According to data from Zhiyan Consulting (Intelligence Research Group), the global analgesic drug market size reached approximately $91.14 billion in 2022, while China's analgesic drug market size amounted to about 122.6 billion yuan. Hyperway Pharma' novel mechanism analgesic HBW-004285 tablets, a Nav1.8 inhibitor, does not produce opioid-like addictive properties and is expected to emerge as one of the game-changers in the traditional analgesics market.
HBW-004285 tablets have entered Phase II clinical trials. Phase I clinical data indicates that compared to VX-548—the world's first approved Nav1.8 inhibitor—HBW-004285 demonstrates superior safety, faster onset of action, optimized pharmacokinetics, and no gender-related differences, positioning it to potentially outperform VX-548.
Bruton's tyrosine kinase (BTK) is a key regulator of B cell receptor signal transduction pathway, which is closely related to a variety of B cell tumors (such as lymphoma, leukemia)
and autoimmune diseases (such as systemic lupus erythematosus, multiple sclerosis). Although only six BTK inhibitors have been approved to market in the world, they have achieved great success in the treatment of lymphoma and leukemia, giving rise to the blockbuster drug ibrutinib with annual sales exceeding $9 billion. And with the application of BTK inhibitors in the field of autoimmune diseases, it is expected that the global market size of BTK inhibitors will reach $26.1 billion by 2030. However, the resistance of the first and second generation BTK inhibitors has inevitably emerged, so scientists have developed non covalent BTK inhibitors that do not rely on the C481 site.
HBW-3220 capsules are poised to initiate Phase III clinical trials. As the world's most rapidly advancing fourth-generation BTK inhibitor, it overcomes multiple drug-resistant mutations (such as C481S, L528W, T474I) developed against the first three generations of BTK inhibitors. Available research data demonstrate HBW-3220's favorable safety profile and significant therapeutic efficacy. It exhibits particularly outstanding results in patients with chronic lymphocytic leukemia (CLL) and small lymphocytic lymphoma (SLL), achieving an objective response rate (ORR) of 100% in CLL/SLL patients harboring the C481S mutation. For CLL/SLL patients who have previously taken irreversible BTK inhibitors and BCL2 inhibitors, its ORR has surpassed Pirtobrutinib, positioning it to potentially overtake competitors and capture the follow-on treatment market for BTK inhibitors valued at tens of billions of dollars.
Additionally, HBW-3220 capsules hold significant development potential in the field of autoimmune diseases, having already received a clinical trial approval for an expanded indication in nephropathy.
KRAS was once considered an “undruggable target.” The KRAS G12D mutation is the most common type of KRAS mutation and occurs more frequently in cancers that are difficult to treat or have poor treatment outcomes, such as pancreatic cancer. Studies indicate that approximately 90% of pancreatic cancer patients harbor KRAS mutations, with KRAS G12D mutations accounting for the highest proportion at 45% (compared to only 1% for G12C mutations). Furthermore, in colorectal cancer—the third most common cancer globally—the G12D mutation accounts for 45% of KRAS-mutated cases. Consequently, developing KRAS G12D inhibitors not only holds significant market potential but also addresses an urgent need to safeguard the health and lives of millions of patients.
Leveraging the capabilities in drug design, synthesis, and comprehensive drugability evaluation, Hyperway Pharma has developed HBW-012336—an orally administered KRAS G12D inhibitor exhibiting high activity, selectivity, tissue targeting, and potent antitumor efficacy. This project received clinical trial approval in April 2025 and is currently undergoing Phase I clinical trials.
KRAS mutations are diverse, encompassing not only G12C and G12D mutations but also over 20 other variants such as G12V, G12R, and G13D. Among these, the seven predominant mutations account for more than 90% of all KRAS mutations. Therefore, developing Pan-KRAS inhibitors capable of broadly suppressing the core spectrum of KRAS mutations has become a hotspot in current drug research and development, with the potential to become one of the most powerful anti-cancer weapons in the future. Estimates suggest that KRAS G12C and KRAS G12D inhibitors alone will constitute a market worth approximately $10 billion. Pan-KRAS inhibitors, however, will overcome the limitations of single-mutation KRAS inhibitors by covering multiple mutation types, overcoming resistance, and offering broader applicability and superior combination effects. Their market value is projected to be eight times that of KRAS G12C inhibitors, attracting numerous pharmaceutical companies to enter the field.
Hyperway Pharma' high-potency, highly selective, and tissue-targeted oral Pan-KRAS inhibitor HBW-012462 simultaneously inhibits both KRAS (ON) and KRAS (OFF) states. Compared to publicly available data on Pan-KRAS inhibitors currently in development globally, HBW-012462 demonstrates superior or comparable performance in key preclinical drug-development metrics including cellular activity, in vivo efficacy, and pharmacokinetics, indicating significant development potential. Clinical studies are currently underway.
The significant increase in the types and number of patients with neurological disorders has created an increasingly urgent global demand for neurological drugs, driving rapid growth in the pharmaceutical market. This market is projected to reach $172.1 billion by 2034. Hyperway Pharma has established a brain-penetrant drug development platform with distinct competitive advantages, focusing on developing brain disease drugs capable of penetrating the blood-brain barrier. The company has deployed multiple innovative drug pipelines and identified several brain-targeted drugs with brain penetration rate exceeding 60%. HBW-3210 Capsule is one such promising candidate.
HBW-3210 Capsule features two major innovations: First, as a fourth-generation non-covalent BTK inhibitor, it overcomes resistance issues caused by the C481S mutation in first and second generation BTK inhibitors, demonstrating high inhibitory activity against both wild-type and mutant BTK kinases. Second, it exhibits potent brain penetration. Compared to Tirabrutinib (developed by Ono Pharmaceutical in Japan)—the world's only approved first-generation irreversible BTK inhibitor for relapsed or refractory PCNSL—HBW-3210 demonstrates superior brain permeability (rat BBB penetration rate: >60% vs ~10%) and efficacy. This demonstrates significant development potential and the possibility of becoming a FIC compound.
Currently, HBW-3210 capsules are undergoing Phase I clinical trials and have demonstrated remarkable therapeutic effects. Notably, a patient who progressed due to resistance to a previously used brain-penetrant BTK inhibitor achieved complete disappearance of brain tumors after six cycles of HBW-3210 capsules, achieving outstanding therapeutic outcomes.
Collaboration
-
 Innovative Medicines
Innovative Medicines
Regional Rights Assignment -
 Innovative Medicines
Innovative Medicines
Collaborative Development -
 Innovative drug
Innovative drug
R&D technology transfer -
 Other Medicinal Products
Other Medicinal Products
Research and Development Collaboration
Innovative drug development is a lengthy and challenging process. As China's pharmaceutical industry undergoes a period of profound transformation, Hyperway Pharma will adopt an open, multi-tiered, and multi-faceted collaboration model to maximize resource integration, concentrate R&D efforts, accelerate drug development, and avoid homogenized competition. Embracing an attitude of “The sea admits hundreds of rivers,” we seek like-minded partners both domestically and internationally to jointly develop high-quality, cost-effective Chinese medicines!